Insulated molecular wires: the smallest electric power cord
28.01.2019
 |
|
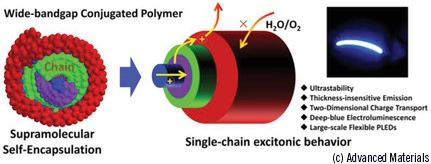
The helical structure of the insulated molecular wire proposed for stable light-emitting applications. Picture credit: Advanced Materials. |
A new supramolecular self-encapsulation strategy provides an effective route to construct stable light-emitting polymers for optoelectronic applications.
Flexible polymer light-emitting devices have numerous potential applications in wearable equipment such as biosmart devices. Soft bioelectronics can provide accurate and real-time sensing of physiological signals such as glucose or blood pressure, and constitute a new paradigm for health monitoring. The basis material of these devices –conjugated polymers–degrades rapidly, affecting the color purity and luminescence efficiency. The fragile equilibrium of conjugated polymers has been the limiting factor for the massive commercialization of plastic electronics, and nowadays the efforts of many research groups have focused on guaranteeing the stability of these organic materials.
The group of Juan Cabanillas (IMDEA Nanociencia), in collaboration with groups at Nanjing Tech University (China) and other research centres have worked on an original strategy to overcome the problem. A supramolecular self-encapsulation of the polymer chain has been proposed for insulating the aromatic backbone from external agents (oxygen, moisture) whilst avoiding the formation of interchain excited states, aggregation, and other obstacles hindering the spectral purity. The conjugated polydiaryl-fluorene (PHDPF-Cz) consists of a mainchain backbone where diphenyl groups localize at the 9-position, and carbazole (Cz) units localize at the 4-position. The π-stacking of aromatic groups at the side of the chain surround the polymeric chain backbone, in analogy with electric power cords in which the metallic wires are insulated by a surrounding plastic casing. The π-stacked units enable intrachain charge transport and simultaneously hinder the interchain π-π interactions in solid state.
The optical properties of PHDPF-Cz films were studied by Cabanillas and Sun at IMDEA Nanociencia by the ultrafast pump-probe technique. The films display very large emission stability, even upon exposing them to hazardous conditions (8h of thermal annealing at 180 ºC in air), as shown by absence of broad green emission characteristic of oxygen-induced defects and aggregation. The absorbance and photoluminescence spectra are insensitive to the chain environment (either solution, gel or solid film) and film thickness, indicating an excellent property for their industrial processing at a large scale. To investigate the transport capability of PHDPF-Cz, a light-emitting diode was built by sandwiching the film between two electrodes. It was found a high hole mobility -several orders of magnitude higher than previously reported for other spin-coated conjugated polymers that is little dependent on the film thickness (in the range 95-175 nm). Also, the blue luminescence was found to be independent from the current voltage applied (4-9 V), constituting a desired feature for the fabrication of optoelectronic devices.
These so-called insulated molecular wires are attracting strong interest for the fabrication of flexible electronic devices. The advantages of such materials are many. They are soluble, thus could be processed as inks for 2D circuit printing. The chemistry of conjugated polymers is very rich, meaning that is possible to tune the emission properties by materials engineering. And they are organic and flexible, so they could potentially be embedded in biological smart devices. A further step in the engineering of these materials would be an alignment of the chains as ordered fibers for enabling a coherent anisotropic charge transport.
 |
|
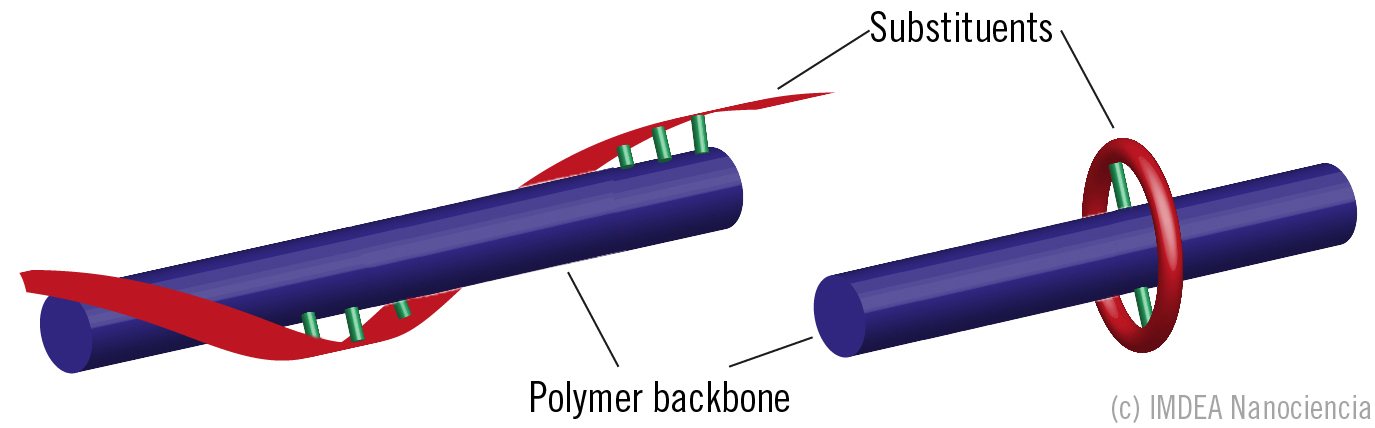
Two strategies for encapsulating π-conjugated polymers by helical-type [1] and ring-type [2] conformation of their own covalently bonded peripheral substituents (green= covalent bonds). |
The group of Cabanillas is currently working on strategies for improving the emission properties of conjugated polymers. Last year, they studied a different strategy for sheathing the backbones by their own side chains with helical or ring-type geometries. “Both strategies work well, but we have proven that helical sheathing strategy [1] forces the co-planar disposition of neighbour monomers, allowing an extended conjugation on which the transport could be much more efficient” Cabanillas says. He continues: “The electronic mobility along the chains is truly higher that the interchain mobility predominant in a conventional conjugated polymer, and this opens many new interesting possibilities for polymer light-emitting materials, including light-amplifying uses”.
Juan Cabanillas is a researcher at IMDEA Nanociencia. His research interests concern with excited state dynamics in conjugated polymers and the application of these materials to different fields such as lighting, light detection and chemical sensing. The work here presented has been co-funded by the Spanish Ministry of Economy and Competitiveness, the China Scholarship Council and the Severo Ochoa Programme for Centres of Excellence.
References:
|
Contact: Dr. Juan Cabanillas González
juan.cabanillas [at] imdea.org
IMDEA Nanociencia Outreach Office
divulgacion.nanociencia [at] imdea.org
+34 91 299 87 12
Twitter: @IMDEA_nano
Facebook: @IMDEANanociencia
|




